Characterizing antibodies with mass photometry
Antibody characterization insights
Mass photometry is a transformative technology for characterizing antibodies during production and offers rapid insights into antigen-antibody interactions, antibody aggregation and fragmentation
Unlock rapid antibody analytics
During antibody manufacturing, efficient and reliable analytics are crucial for optimizing your antibody product. In-process analytics are especially valuable, particularly when interrogating the affinity of an antibody for its intended target, or when assessing the purity and stability of an antibody product. Traditional analysis methods can fall short of the mark when it comes to in-process analytics, often requiring a time-consuming workflow and substantial sample consumption to gain any valuable insight into the antibody sample.
Mass photometry is a novel analytical technique that analyzes antibody samples by determining the molecular mass distribution of antibodies at the single-molecule level. It can resolve antigen-antibody interactions, antibody aggregation, and fragmentation – all with minimal sample consumption and in a turnaround time of just a few minutes.
Delivered by the TwoMP mass photometer, the technique is ideal for testing antibody samples at various stages during the production process, quickly delivering up-to-date data on affinity and sample integrity, with minimal disruption. Mass photometry can also quantify interactions of individual antibody molecules with target molecules.
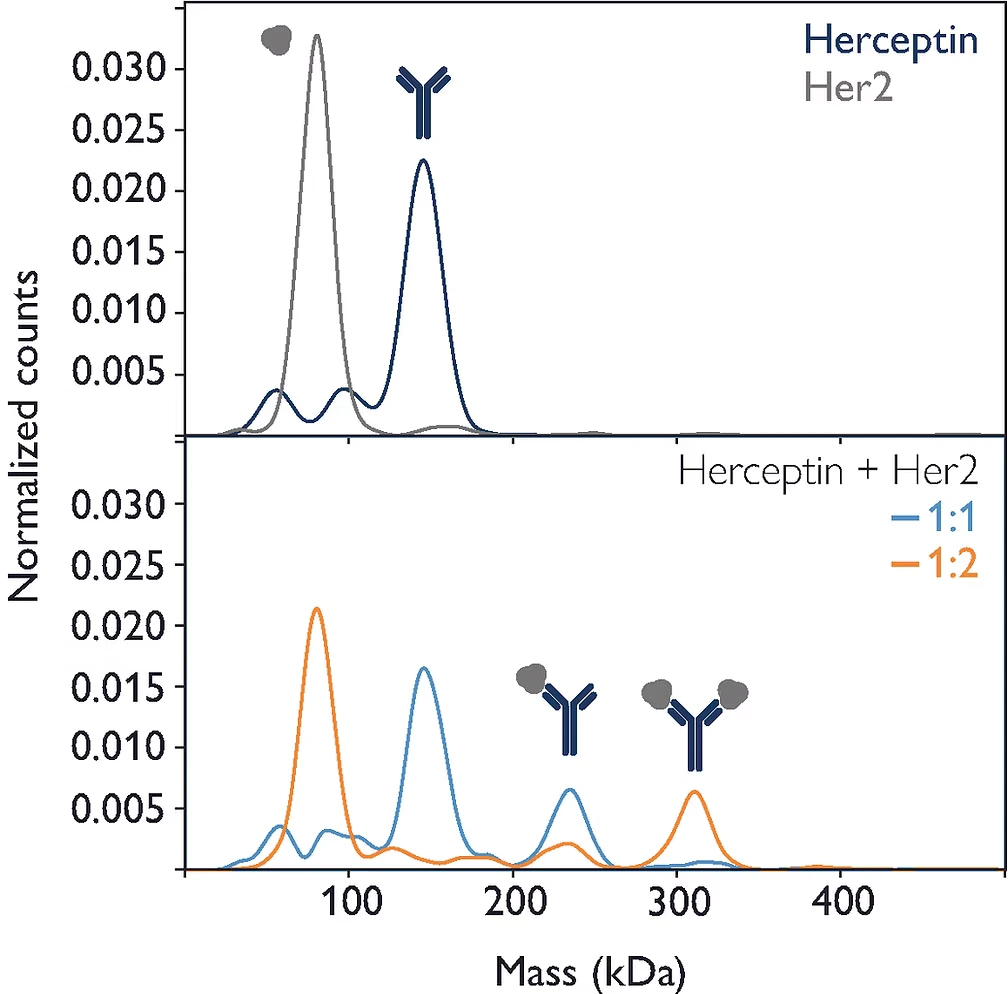
Figure 1: The monoclonal antibody Herceptin and its target, Her2, were measured individually and in mixtures, demonstrating the instrument’s ability to quantify the interactions of individual antibody molecules with target molecules.
Resolve antibody aggregation and fragmentation
Aggregation and fragmentation can significantly undermine the efficacy and safety of antibody-based products. Being able to detect these issues at various stages of product development is crucial for maintaining high-quality therapeutics. In addition to aggregation and fragmentation events, forced degradation studies are also routinely conducted to evaluate the stability of antibodies, ensuring that they perform as intended even under stress.
Mass photometry presents a sensitive and convenient solution for analyzing these key critical quality attributes of antibodies. It can detect contaminants within antibody samples down to picomolar concentrations, operating effectively across a broad range of buffers and experimental conditions. The technique also integrates seamlessly with other analytical techniques such as Size Exclusion Chromatography with Multi-Angle Light Scattering (SEC-MALS).
To further improve turnaround times in antibody analytics, Refeyn provides the Antibody Stability Software Module – which automatically quantifies aggregation in large sample batches in a fraction of the time required for manual analysis, providing statistical analysis for replicates.
The strengths of mass photometry make it a game-changer for fast, in-process analysis of sample purity and stability, transforming the landscape of antibody analytics by providing convenience, ensuring quality and accelerating development.
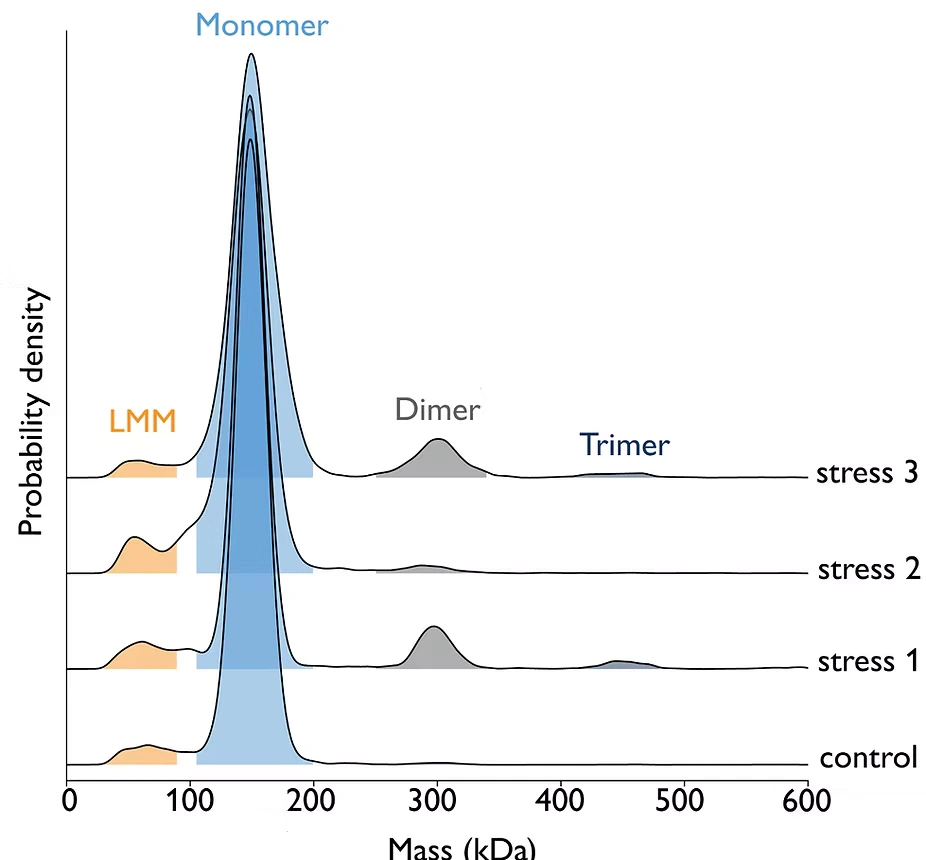
Figure 2: Exposure of antibody samples to three different stress conditions (light, pH change, or peroxide). Samples were measured at 10 nM concentration using 5 μl on the TwoMP Auto. Results show that stress conditions 1 and 3 promote aggregation (specifically, the formation of dimers and trimers), relative to the control sample.
Reveal antigen-antibody interactions and more
Mass photometry excels in revealing critical insights into antibody-antigen interactions and a host of additional antibody dynamics. This sensitive and high-precision technique detects both individual elements and complexes within samples, even identifying low-abundance components (<1%). This capability allows for the rapid determination of the dissociation constant (Kd) of antigen-antibody interactions, providing essential data on the affinity of the antibodies produced.
Enhancing the capabilities of mass photometry, the MassFluidix HC microfluidics add-on for the TwoMP mass photometer enables the characterization of extremely low-affinity interactions. By rapidly diluting the sample prior to measurement, it exposes complexes that typically remain undetected at the standard nanomolar concentrations used in mass photometry. This approach offers a deeper understanding of antibody behavior, driving more robust and effective antibody development.
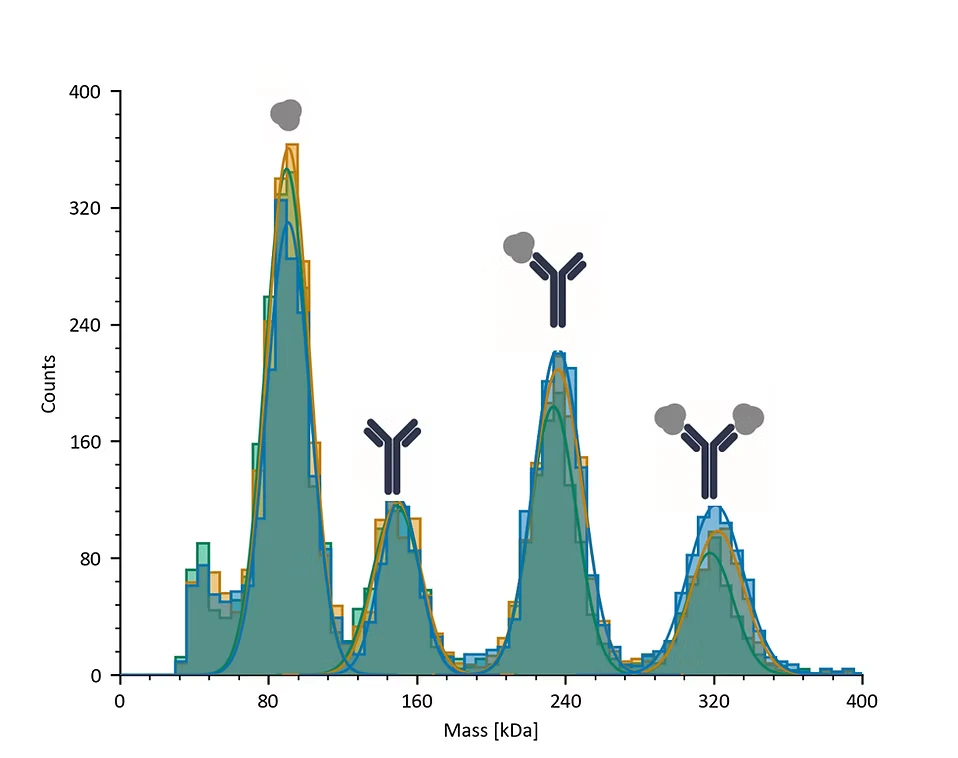
Figure 3: Mass histograms of a triplicate mass photometry measurement of a therapeutic antibody mixed with EGFR at a ratio of 2:1. Mass photometry detected each component separately as well as in complexes of different stoichiometries. The data from the measurements was used to estimate the binding affinities of the antibody:EGFR complexes (1:1=7.0 ± 2.0 nM; 1:2=25.3 ± 6.8 nM).
Mass photometry solutions for antibody analysis
- TwoMP
-

For molecular mass measurements with unmatched sensitivity, speed and simplicity of use, a TwoMP mass photometer offers a wide mass range and single-molecule resolution.
High-fidelity measurements of molecular mass
Little sample required
Intuitive acquisition and data analysis software
Easy setup – a compact, benchtop instrument with minimal installation requirements
- TwoMP Auto
-

The automated mass photometer frees up operator time and offers enhanced precision, enabling rapid measurement of multiple samples with low sample consumption.
Rapid, automated measurement of multiple samples
Ideal for screening and titration assays
Highly reproducible data with user-friendly software
- MassFluidix HC
-

Refeyn’s microfluidics system, MassFluidix HC, significantly expands the range of sample concentrations amenable to investigation by mass photometry, by raising the upper sample concentration limit from the nanomolar to the micromolar range. This enables applications such as the characterization of low-affinity interactions.
- Enables measurement of concentrated samples
- Uses rapid dilution
- Is an add-on for TwoMP and OneMP mass photometers
- Refeyn consumables
-
 By using Refeyn consumables, you can spend less time preparing for measurements and gain greater confidence in your data. The consumables range includes calibrants, sample carrier slides and more.
By using Refeyn consumables, you can spend less time preparing for measurements and gain greater confidence in your data. The consumables range includes calibrants, sample carrier slides and more. - Antibody Stability Module
-
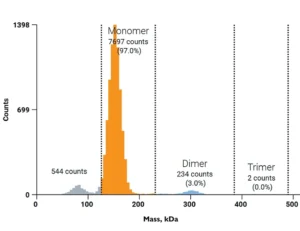
This software module characterizes aggregation in large batches of antibody samples in a fraction of the time required for manual analysis. It supports grouping by replicate and antibody type, and can distinguish and quantify different oligomeric species based on the expected monomer mass.

For molecular mass measurements with unmatched sensitivity, speed and simplicity of use, a TwoMP mass photometer offers a wide mass range and single-molecule resolution.
High-fidelity measurements of molecular mass
Little sample required
Intuitive acquisition and data analysis software
Easy setup – a compact, benchtop instrument with minimal installation requirements

The automated mass photometer frees up operator time and offers enhanced precision, enabling rapid measurement of multiple samples with low sample consumption.
Rapid, automated measurement of multiple samples
Ideal for screening and titration assays
Highly reproducible data with user-friendly software

Refeyn’s microfluidics system, MassFluidix HC, significantly expands the range of sample concentrations amenable to investigation by mass photometry, by raising the upper sample concentration limit from the nanomolar to the micromolar range. This enables applications such as the characterization of low-affinity interactions.
- Enables measurement of concentrated samples
- Uses rapid dilution
- Is an add-on for TwoMP and OneMP mass photometers


This software module characterizes aggregation in large batches of antibody samples in a fraction of the time required for manual analysis. It supports grouping by replicate and antibody type, and can distinguish and quantify different oligomeric species based on the expected monomer mass.
Testimonials from mass photometry users
Radic et al. (2023)
Buffington et al. (2023)
“Mass photometry fits perfectly into our analytical workflows when we know there are additional species in our samples [such as aggregates] but we don’t know their mass or their oligomeric state. Now we finally have a suitable technology to fill that gap.”
Dr. Martin E
experienced scientist at one of the top 5 pharmaceutical companies
View further resources related to antibody characterization
- Selected publications
-
Claasen, M., Kofinova, Z., Contino, M., Struwe, W.B., 2024. Analysis of Protein Complex Formation at Micromolar Concentrations by Coupling Microfluidics with Mass Photometry. Journal of Visualized Experiments. (203), e65772. [Link]
Radić, L., Sliepen, K., Yin, V., Brinkkemper, M., Capella-Pujol, J., Schriek, A.I., Torres, J.L., Bangaru, S., Burger, J.A., Poniman, M. and Bontjer, I., 2023. Bispecific antibodies combine breadth, potency, and avidity of parental antibodies to neutralize sarbecoviruses. Iscience, 26(4). [Link]
Yin, V., Lai, S.H., Caniels, T.G., Brouwer, P.J., Brinkkemper, M., Aldon, Y., Liu, H., Yuan, M., Wilson, I.A., Sanders, R.W. and Van Gils, M.J., 2021. Probing affinity, avidity, anticooperativity, and competition in antibody and receptor binding to the SARS-CoV-2 spike by single particle mass analyses. ACS central science, 7(11), pp.1863-1873. [Link]
den Boer, M.A., Lai, S.H., Xue, X., van Kampen, M.D., Bleijlevens, B. and Heck, A.J., 2021. Comparative analysis of antibodies and heavily glycosylated macromolecular immune complexes by size-exclusion chromatography multi-angle light scattering, native charge detection mass spectrometry, and mass photometry. Analytical Chemistry, 94(2), pp.892-900. [Link]
Soltermann, F., Foley, E.D., Pagnoni, V., Galpin, M., Benesch, J.L., Kukura, P. and Struwe, W.B., 2020. Quantifying protein–protein interactions by molecular counting with mass photometry. Angewandte Chemie International Edition, 59(27), pp.10774-10779. [Link]
Wu, D. and Piszczek, G., 2020. Measuring the affinity of protein-protein interactions on a single-molecule level by mass photometry. Analytical biochemistry, 592, p.113575. [Link]
- App notes & Tech notes
-
App note: Assessing protein samples by mass photometry and size exclusion chromatography
App note: Characterization of forced antibody degradation with automated mass photometry
Tech note: Streamline antibody stability analysis with mass photometry
- Comparing mass photometry and SEC for antibody aggregation assessment
- Brochures and eBooks
-
- Webinars
-
- Refeyn posts
-
Claasen, M., Kofinova, Z., Contino, M., Struwe, W.B., 2024. Analysis of Protein Complex Formation at Micromolar Concentrations by Coupling Microfluidics with Mass Photometry. Journal of Visualized Experiments. (203), e65772. [Link]
Radić, L., Sliepen, K., Yin, V., Brinkkemper, M., Capella-Pujol, J., Schriek, A.I., Torres, J.L., Bangaru, S., Burger, J.A., Poniman, M. and Bontjer, I., 2023. Bispecific antibodies combine breadth, potency, and avidity of parental antibodies to neutralize sarbecoviruses. Iscience, 26(4). [Link]
Yin, V., Lai, S.H., Caniels, T.G., Brouwer, P.J., Brinkkemper, M., Aldon, Y., Liu, H., Yuan, M., Wilson, I.A., Sanders, R.W. and Van Gils, M.J., 2021. Probing affinity, avidity, anticooperativity, and competition in antibody and receptor binding to the SARS-CoV-2 spike by single particle mass analyses. ACS central science, 7(11), pp.1863-1873. [Link]
den Boer, M.A., Lai, S.H., Xue, X., van Kampen, M.D., Bleijlevens, B. and Heck, A.J., 2021. Comparative analysis of antibodies and heavily glycosylated macromolecular immune complexes by size-exclusion chromatography multi-angle light scattering, native charge detection mass spectrometry, and mass photometry. Analytical Chemistry, 94(2), pp.892-900. [Link]
Soltermann, F., Foley, E.D., Pagnoni, V., Galpin, M., Benesch, J.L., Kukura, P. and Struwe, W.B., 2020. Quantifying protein–protein interactions by molecular counting with mass photometry. Angewandte Chemie International Edition, 59(27), pp.10774-10779. [Link]
Wu, D. and Piszczek, G., 2020. Measuring the affinity of protein-protein interactions on a single-molecule level by mass photometry. Analytical biochemistry, 592, p.113575. [Link]
App note: Assessing protein samples by mass photometry and size exclusion chromatography
App note: Characterization of forced antibody degradation with automated mass photometry
Tech note: Streamline antibody stability analysis with mass photometry
- Comparing mass photometry and SEC for antibody aggregation assessment

