Mass Photometry for Antibody Stability and Aggregation Analysis
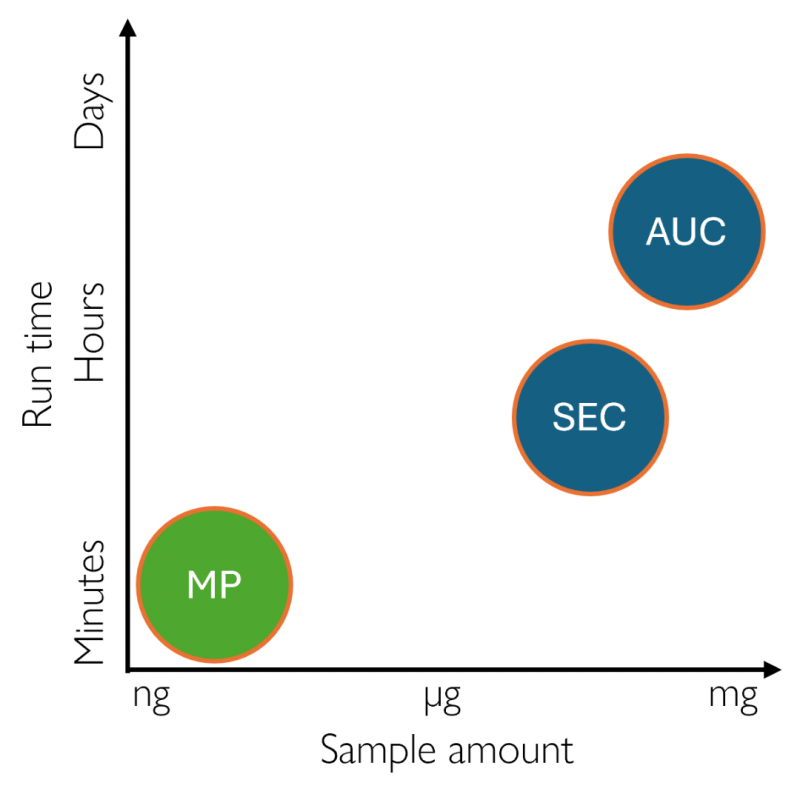
Measure antibody purity, stability, and aggregation faster and with less sample than SEC or AUC
Column-free antibody aggregation characterization
From monoclonal antibodies (mAbs) to advanced modalities like bispecifics and ADCs, antibodies are powerful therapeutics. Antibody characterization and aggregation measurement are critical steps in therapeutic antibody discovery and development. Robust aggregation analysis is critical for ensuring safety, efficacy, and stability – particularly for complex molecules. Refeyn’s column-free mass photometry technology measures aggregation fast, with no method optimization and minimal sample, guiding formulation and process development.
Key pain points with traditional aggregation analysis methods:
Time-consuming method development across different modalities
Column interactions can distort results
High sample consumption
Slow processing
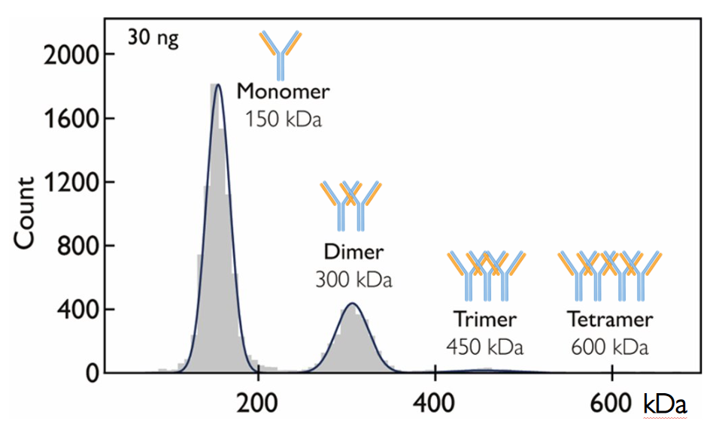
Figure 1. Mass photometry resolves and quantifies aggregate species directly in solution. In this example, 30 ng of sample was used. Mass photometry measurements take one minute.
Challenges
Complex method development for antibody analytics
Traditional column-based methods, such as size-exclusion chromatography (SEC), require optimization for different antibody modalities (mAbs, BsAbs, ADCs and AOCs). Column type (matrix) and buffer composition frequently need to be tailored for each modality, and even for different samples, slowing analytical development and process development workflows.
Solution: Mass photometry requires no method optimization
Mass photometry eliminates this bottleneck. It is a universal antibody analytics technique that works the same for mAbs, bispecifics and other complex modalities – with no method development, using minimal sample and providing results in minutes. Across modalities, mass photometry consistently delivers reliable therapeutic antibody aggregation data – with accuracy benchmarked against SEC and analytical ultracentrifugation (AUC). It resolves different aggregate species (dimers, trimers, and higher-order forms) with baseline resolution, making it clear exactly which species are present in every sample across the antibody development lifecycle.
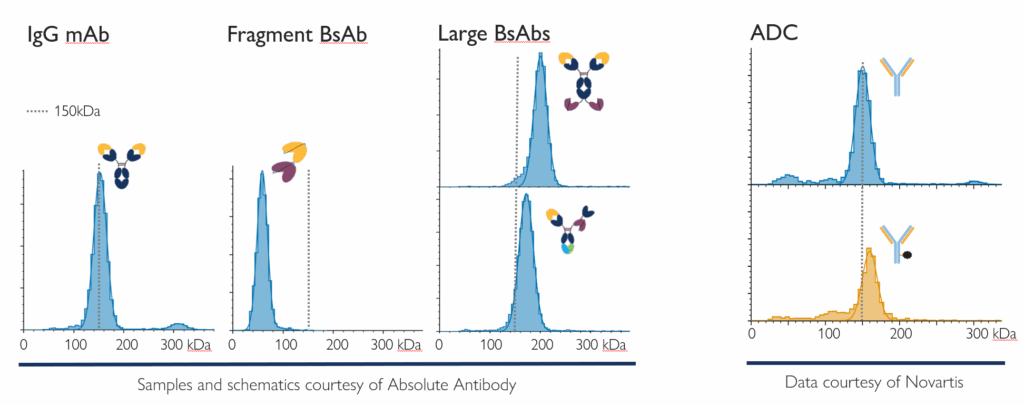
Figure 2. Mass photometry works for different antibody modalities with no need for method development: A monoclonal antibody (mAb), different bispecific formats, and an ADC (non-conjugated and conjugated). Each measurement shown used just nanograms of sample diluted in PBS buffer. Through single-molecule mass measurement, it directly measures aggregation, fragmentation and binding.
Column interactions can distort aggregation results
In therapeutic antibody characterization, column-based methods like SEC can introduce artifacts. Antibody molecules can interact with the column matrix, altering sample properties and retention time, complicating aggregation data interpretation. This issue becomes more pronounced for more complex antibody modalities, such as bispecifics and ADCs.
Different modalities show different interaction risks. For example, ADCs are more prone to ‘sticking’ to columns due to their hydrophobic payloads, often requiring salt and buffer optimization. Some modalities can partially adhere to columns, leading to fragmentation or loss of material – and again requiring further optimization of matrix and buffer conditions.
Solution: Mass photometry preserves true aggregation profiles
Mass photometry avoids column-based artifacts entirely. With no need for a column, there are no unexpected interactions. It measures therapeutic antibody aggregation directly in solution, preserving native species and ensuring accurate, unbiased results across mAbs, bispecifics, ADCs, and other emerging modalities.
Mass photometry detects dimers, trimers, and higher-order aggregates with high resolution – even for challenging modalities – delivering reliable data without sample loss, method optimization, or the risk of column-induced structural changes. It provides clear insights into aggregation as a critical quality attribute.
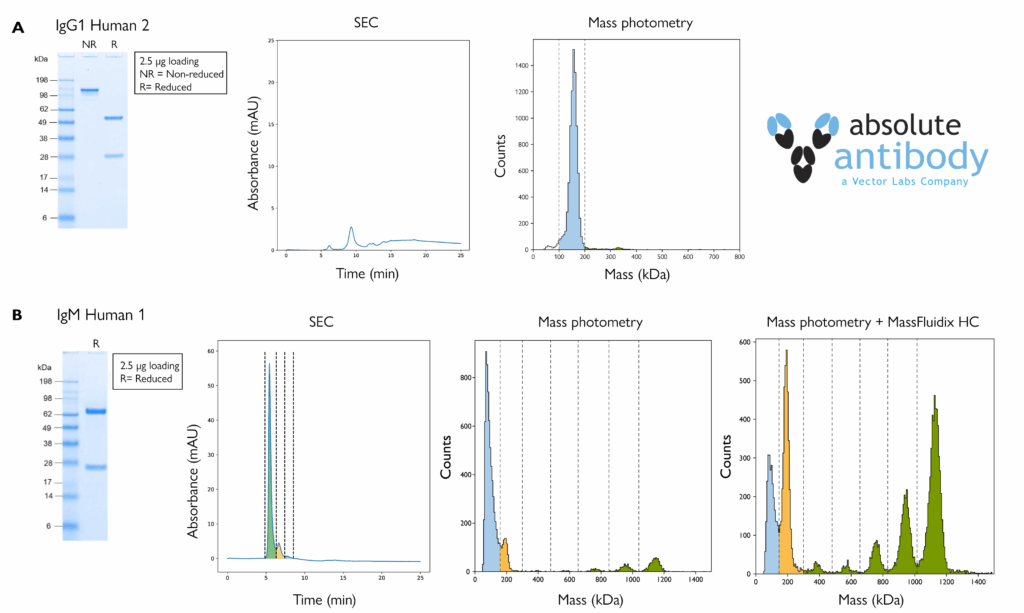
Figure 3. A) Mass photometry, but not SEC, returned clear aggregation readouts for an IgG antibody. Data is shown for analysis of antibody IgG1 Human 2 by SDS-PAGE run under non-reducing (NR) and reducing (R) conditions (left), SEC-UV (middle) and mass photometry (right). The SEC profile suggests that much of the sample was retained within the column. B) IgM pentamers and hexamers were only resolved by mass photometry. Data is shown for analysis of antibody IgM Human 1 by SDS-PAGE run under reducing (R) conditions (far left), SEC (mid left), standard mass photometry (mid right) and mass photometry with MassFluidix HC (far right). The colored peak regions correspond to monomers (blue), dimers (light orange) and higher-order species (green).
High sample consumption
Traditional aggregation analysis methods can require large amounts of valuable therapeutic antibody material. SEC typically operates on the order of micrograms, while AUC can require hundreds of micrograms per sample. For early-stage antibody candidates in particular – including mAbs, bispecifics and ADCs – high sample consumption can limit throughput and slow down development.
Solution: Mass photometry uses little sample but delivers high-accuracy data
Mass photometry delivers accurate aggregation measurements using only nanograms of sample, preserving precious antibody material. This makes it ideal for early-stage discovery, process development, and characterization workflows across all antibody modalities. Despite using minimal sample, mass photometry reliably detects and quantifies dimers, trimers, and higher-order aggregates, with high resolution and accuracy benchmarked against SEC and AUC.
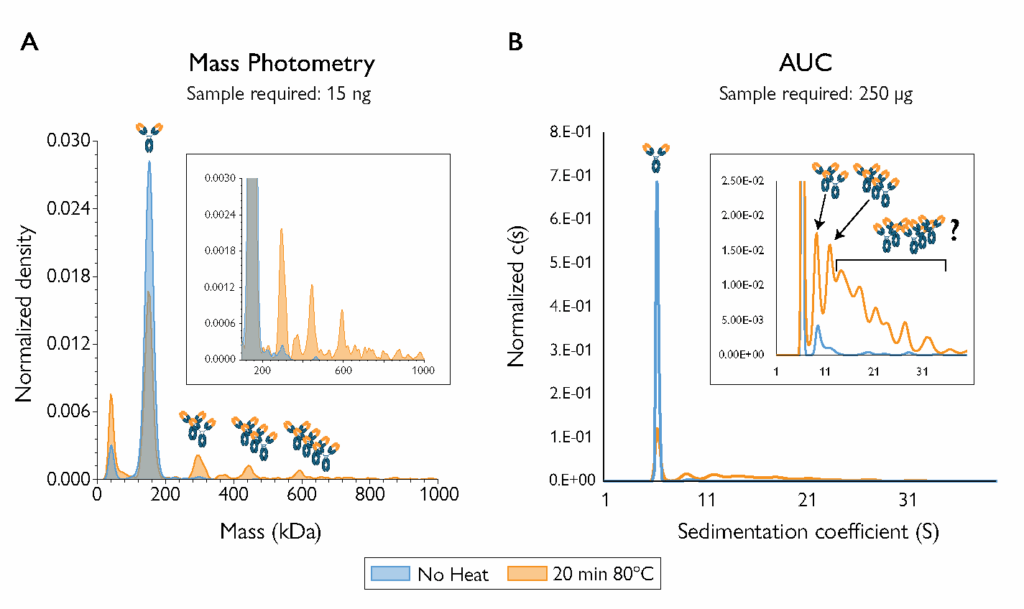
Figure 4. Mass photometry resolved more aggregate species than AUC in heat-stressed samples of the NISTmAb standard, and used 10,000x less sample. The AUC measurement used 250 µg; mass photometry used 15 ng.
Traditional aggregation workflows can be time-consuming, delaying decisions in therapeutic antibody development. Standard SEC runs typically take ~20 minutes per sample (without accounting for method optimization time), while AUC requires extensive sample preparation and can then take 4+ hours to complete. These long run times slow characterization and process optimization across mAbs, bispecifics, ADCs, and other emerging modalities.
Solution: Mass photometry delivers results in minutes
Mass photometry provides rapid, high-resolution aggregation measurements in less than five minutes (from sample prep to analysis), and involves no method development and minimal sample preparation. This rapid measurement capability supports efficient screening of antibody candidates and faster process development decisions. Across antibody modalities, mass photometry enables accelerated aggregation analytics without compromising accuracy or sensitivity.
Figure 5. Mass photometry measurements take minutes and use nanograms of sample, while AUC and SEC take much longer (with sample preparation and method development demanding even more time) and use significantly more sample.
Mass photometry solutions for antibody aggregation analysis
Refeyn TwoMP & TwoMP Auto
The TwoMP provides unique insight into antibody samples by measuring the mass of individual molecules in native conditions label-free and with single-molecule resolution.

It enables accurate, multi-attribute characterization of antibody therapeutics, including complex modalities, with minimal sample preparation and nanograms of sample. Designed for intuitive operation, it delivers reliable results within minutes.
The TwoMP Auto extends these capabilities with automated handling for up to 24 measurements – increasing throughput, improving repeatability, and offering walkaway time.
Its core features address key analytical challenges in antibody aggregation:
Minimal Sample Requirements
Requires only nanograms of sample for analysis, preserving precious material.
Rapid Analysis
Delivers comprehensive antibody aggregation data within minutes per sample.
Native Single-Molecule Precision
Utilizes single-molecule resolution to accurately measure molecular mass under native conditions.
MassFluidix™ HC System
Refeyn’s microfluidics system, MassFluidix HC, raises the upper sample concentration limit from the nanomolar to the micromolar range.
Significantly expanding the sample concentration range amenable to investigation by mass photometry, MassFluidix HC is an add-on for Refeyn’s TwoMP mass photometer. It includes a central unit with fluid controls. Ready-to-use rapid-dilution microfluidics chips are also available from Refeyn for this system.
MassFluidix offers:
Characterization of concentrated samples
Starting concentration up to the tens of micromolar
Rapid analysis requiring little sample
~20 min for setup and cleaning, using 20–60 µL of sample
User-friendly operation
User training takes just one day, supported by intuitive fluid-control software
MassGlass™ UC Slides & MassFerence™ P1 Calibrant

MassGlass™ UC Slides
High-quality mass photometry measurements begin with clean slides, and MassGlass UC provide a pristine surface, straight out of the box.
Refeyn offers MassGlass UC – Sample Prep Kits for the TwoMP and TwoMP Auto systems, containing everything you need for a seamless experience when measuring your antibody samples.
MassFerence™ P1 Calibrant
MassFerence P1 enables accurate, reproducible protein
mass calibration across 90 – 1000 kDa for OneMP, TwoMP,
and TwoMP Auto.
AcquireMP acquisition and DiscoverMP analysis software
Refeyn’s AquireMP acquisition and DiscoverMP analysis software provides an intuitive, high-performance solution for single-molecule mass photometry workflows.
Designed to streamline both data capture and interpretation, the software suite enables real-time measurement with clear user guidance and powerful analysis tools – making it an essential companion for researchers working with complex samples.
Its core features support fast, confident decision-making:
User-Friendly Interface
Intuitive interface with minimal parameters to optimize.
Real-Time Quality Control
Real-time results and quality metrics ensure high-quality data capture.
Instant Report Generation
Advanced analysis and graphing tools deliver report-ready insights in minutes.
Refeyn’s Antibody Stability software module characterizes aggregation in large batches of antibody samples in a fraction of the time required for manual analysis. It supports grouping by replicate and antibody type, and can distinguish and quantify different oligomeric species based on the expected monomer mass.
Mass photometry for antibody analysis
Mass photometry vs. SEC: Quick overview
Compared to SEC, mass photometry is faster, avoids column interaction issues, and uses less sample, as explained by Refeyn Market Development Manager Salma Jalal, PhD.
Frequently asked questions
How long do antibody aggregation measurements take using mass photometry?
Mass photometry delivers results in under 5 minutes, from sample prep to analysis The measurement itself takes only 1 minute. This rapid measurement capability enables quicker, data-driven decisions throughout antibody development processes and manufacturing workflows. By comparison a typical SEC run is ~20 minutes and AUC takes 4+ hours.
How does mass photometry measure antibody aggregation?
Mass photometry directly measures the mass of individual molecules in their native state, label-free and with single-molecule resolution, and uses the principles of interferometric scattering microscopy. To learn more, read the article, How Mass Photometry Works, or watch the video, Mass Photometry 101.
How much sample is required for antibody aggregation analysis with mass photometry?
A mass photometry measurement uses 10-20 µL of sample with 1-10 nM antibody concentration – amounting to 15–30 ng. This makes mass photometry ideal for early-stage candidates or studies where sample is limited. In comparison, SEC requires micrograms and AUC requires tens or hundreds of micrograms.
What sample concentrations are used for mass photometry measurements?
For standard mass photometry measurements, samples should be at a nanomolar concentration (1–10 nM). To measure more concentrated samples – for example to assess reversible vs. irreversible aggregates – the, MassFludix HC add-on, a microfluidics system, can be used. It can analyze samples with a concentration up to the tens of micromolar.
Why is mass photometry described as a column-free and label-free technique?
Mass photometry is described as a column‑free and label‑free technique because the measurement is done in solution, with no need for column separation or other sample modifications, such as labelling. This reduces the risk of interactions or artifacts that can arise from using columns or tags. It also means that mass photometry requires no method optimization – it can be used with an array of different buffers.
Can mass photometry analyze bispecific antibodies and ADCs?
Yes. Mass photometry works across all antibody modalities, including monoclonals, bispecifics, multispecifics and ADCs. Its native-state, column‑free, label-free measurement ensures reliable characterization of aggregation.
In fact, as mass photometry simply measures molecular mass, it can be applied to a wide range of samples for multiple uses. For example, it can also analyze antibody-antigen binding, characterize mRNA samples and check sample quality prior to analysis by methods such as cryo-EM.
What are the benefits of the TwoMP Auto system?
The TwoMP Auto enables rapid screening of up to 24 conditions in as little as 90 minutes, helping accelerate antibody manufacturing workflows. Early detection of antibody aggregation issues allows for earlier adjustments, reducing development bottlenecks and improving overall process efficiency.
How does mass photometry compare to AUC and SEC for antibody aggregation analysis?
Mass photometry provides rapid, column-free antibody aggregation analysis in under 5 minutes using only nanograms of sample – making it up to 300x faster and 8,000x more sample‑efficient than AUC. Column‑free and orthogonal to SEC, it eliminates the need for method development and provides accurate, high-resolution measurements using minimal material.
What are the key benefits of using mass photometry for antibody aggregation analysis?
- Mass photometry offers column-free aggregation measurements
- It works reliably across mAbs, bispecifics, ADCs, and other antibody modalities.
- It provides label-free, native-state measurement at the single-molecule level.
- Results are generated rapidly, in just minutes.
- Only nanogram quantities of sample are required for analysis.
- As it is column free, it eliminates column interactions and the need for method optimization.
- It delivers reliable, high-resolution aggregation data, benchmarked against SEC and AUC.
Can mass photometry be used with more concentrated samples (above nanomolar)?
Yes, mass photometry can analyze samples with concentrations up to the tens of micromolar, if the rapid-dilution microfluidics add-on MassFluidix HC is used.
Resources

Characterization of forced antibody degradation with automated mass photometry
Antibody aggregation is a critical concern in the development of biopharmaceuticals, including therapeutic antibodies such as monoclonal antibodies, bispecific antibodies, and antibody-drug conjugates. Mass photometry is a cutting-edge analytical technique that can be used for the characterization of antibody aggregation and degradation, in solution.

Monitoring aggregation levels of biosimilar mAbs using mass photometry and SEC
Learn how mass photometry compares with size-exclusion chromatography in measuring aggregation levels of monoclonal antibodies. Discover the pros and cons of each technology and the factors that should be considered when analyzing and comparing data generated by these different techniques.

Comparing mass photometry and SEC for antibody aggregation assessment
In this app note, a comparison to size-exclusion chromatography (SEC) shows the two techniques agree, but mass photometry has superior resolution, particularly for larger modalities. Mass photometry requires no optimization beyond sample dilution and readily characterized samples that could not initially be measured with SEC. In addition, mass photometry requires >100x less sample and is 20x faster than SEC, and provides an informative mass readout.

Therapeutic antibody stability – a rapid method for forced antibody degradation studies
Forced antibody degradation studies are vital for evaluating aggregation and fragmentation susceptibility – critical factors in biotherapeutic development. In this webinar, explore how mass photometry enables rapid, precise characterization of antibody stability under various conditions. See how mass photometry offers analysis in diverse buffers with minimal sample requirements and how it compares to SEC.

Streamline antibody stability analysis with mass photometry
In this technical note, you will see how to use mass photometry and the Antibody Stability Module to quickly detect and quantify antibody aggregation. It also shows how mass photometry can measure antibody aggregation at both nanomolar and micromolar concentrations using the MassFluidix HC add-on.

A Modern Tool for Next-Gen Therapeutics
In this e-book, we present case studies demonstrating the capabilities of mass photometry (MP) for antibody aggregation (a critical quality attribute, CQA) and binding assessment. The data showcase the strengths of MP for antibody characterization, and how it compares to frequently used techniques, like size-exclusion chromatography (SEC).
Learn more how mass photometry accelerates antibody aggregation analysis
Ready to revolutionize your research?
Find out how mass photometry can transform your sample characterization. Whether you’re focused on drug development, discovery science, or manufacturing, Refeyn’s innovative technology provides the insights you need to accelerate your breakthroughs.
If you have any other questions or would like further details, please get in touch!